QMC simulations reveal magnetic properties of titanium oxide material
By running computationally intensive quantum Monte Carlo simulations at the Argonne Leadership Computing Facility (ALCF), a U.S. Department of Energy (DOE) Office of Science User Facility, researchers have demonstrated the ability to accurately calculate the magnetic properties of a titanium oxide material that exhibits useful properties for renewable energy and computing technologies.
Your house is likely littered with titanium dioxide (TiO2), from the paint on your walls, to the sunscreen and toothpaste in your bathroom, to the papers on your desk. A brilliant white powder, titanium dioxide is a transition metal oxide that has many favorable properties including optical and catalytic ones.
“Titanium oxides are versatile transition metal compounds that may be used for a range of applications, including electronic devices and photocatalysis,” said Olle Heinonen, materials scientist at Argonne National Laboratory.
While titanium dioxide may be the most well-known titanium oxide material, one of its derivatives, Ti4O7, is another material of interest due to its potential applications in resistive memories and oxide-based fuel cell electrodes. To explore such applications, scientists need to better understand its electronic and magnetic properties.
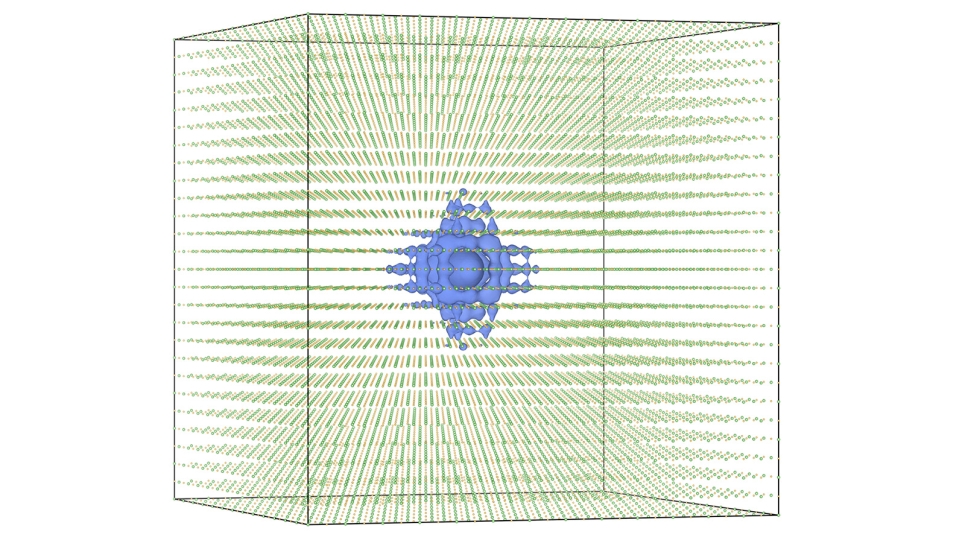
With the computational power of Mira, the ALCF’s 10-petaflops IBM Blue Gene/Q supercomputer, researchers have, for the first time, accurately calculated the magnetic properties of Ti4O7 with quantum Monte Carlo (QMC) simulations.
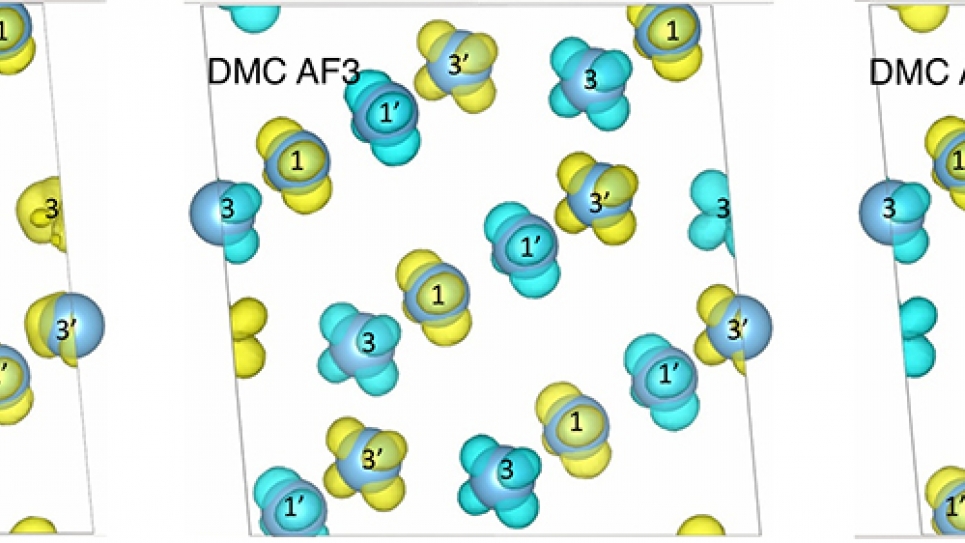
The team’s results, published in Physical Chemistry Chemical Physics, reveal the Ti4O7 ground state—the material’s properties at the lowest possible energy state. By accurately computing the ground state, researchers are able to determine or infer many important material properties, such as crystal structure, conductivity, and magnetism.
“Calculating the ground state is crucial for computational predictions of a material’s behavior in realistic conditions in which temperature, pressure, and time can change its structure,” said Anouar Benali, ALCF computational scientist and lead author of the study.
Because Ti4O7 has several magnetic states close in energy, scientists were previously unable to conclusively determine the ground state through experiment or other computational methods, such as density functional theory (DFT). However, with access to Mira, the research team was able to perform QMC calculations that helped settle the long-standing uncertainty with Ti4O7 by identifying the three magnetic phases that compromise the material’s ground state.
Although QMC requires as much as 1,000 times the computing power of a typical DFT calculation, the method is able to accurately calculate the complex interactions between many electrons. Due to its computational expense, QMC simulations were once limited to modeling systems of small atoms or molecules, but the emergence of supercomputers like Mira has now made it possible to use QMC for rigorous calculations on more complicated materials.
For the Ti4O7 study, researchers used the QMCPACK application developed by Argonne, Oak Ridge, Sandia, and Lawrence Livermore national laboratories. By rewriting the most computational intensive parts of QMCPACK using compiler specific extensions (called vector intrinsics) to better utilize the IBM Blue Gene/Q processor, ALCF computational scientists, including Benali, Ye Luo, and Vitali Morozov, were able to improve QMCPACK performance on Mira by 30 percent. Additionally, by rewriting code to use single precision instead of double precision in key data structures, they decreased the amount of data that needed to be stored in memory by 45 percent.
“These code improvements allowed us to study larger electronic systems in a shorter amount of time,” Benali said.
Computing time at the ALCF was awarded through the Innovative and Novel Computational Impact on Theory and Experiment (INCITE) program. This research was also supported by the DOE Office of Science.
Authors include Benali and Xiaoliang Zhong of Argonne, Heinonen of Argonne and Northwestern University, Luke Shulenburger of Sandia National Laboratories, and Jaron T. Krogel and Paul R. C. Kent of Oak Ridge National Laboratory.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC for the U.S. Department of Energy's Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit the Office of Science website.