Vibrational and Optical Spectroscopy of Electrolyte/Solid Interfaces
Several federal funding agencies have adopted a strategy of sustained fundamental research to address the national energy problem. This project contributes to this strategy by tackling fundamental scientific issues underlying the key question of how best to take advantage of energy from the sun, e.g., through the formation of fuel from the oxidation and reduction of water.
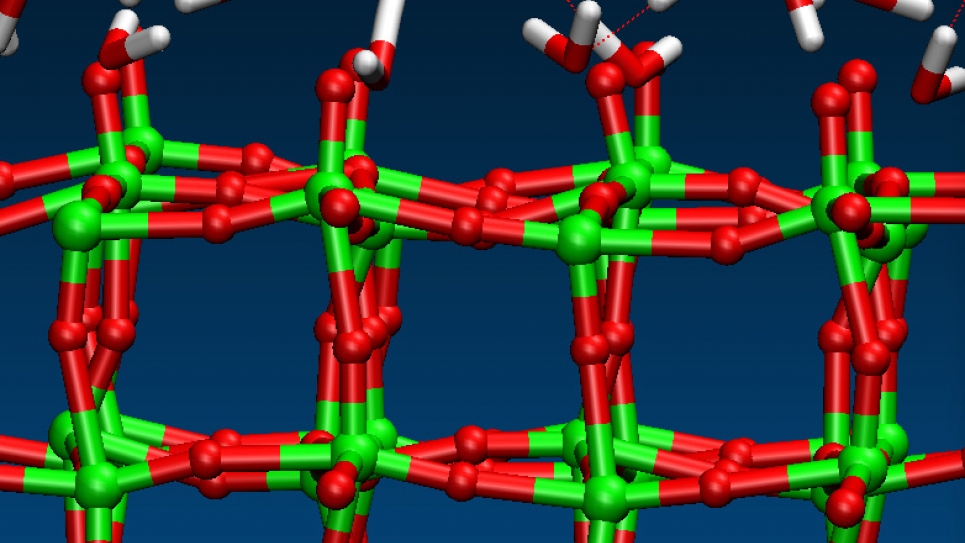
Researchers from the University of California, Davis, and The University of Chicago will use large-scale quantum simulations to model, at the microscopic scale, the physical and chemical processes involved in photo-electrochemical (PEC) energy conversion. The project focuses on processes that occur at the interface between solid photo-electrodes and electrolytes (water with dissolved salts, acids, and bases). Although this project is focused on PEC, the methodologies and processes studied here from first principles are also relevant to other areas of interest to the energy problem, including electrical energy storage.
This project encompasses ab initio molecular dynamics simulations to obtain atomic trajectories, and compute ensemble averages of thermodynamic properties, and calculations, from first principles, of vibrational and electronic spectra. The simulations will rely on codes that have been optimized to take advantage of Mira’s architecture, in particular the Qbox code, and that have shown to exhibit excellent parallel performance.
Researchers aim to provide the knowledge and computational tools necessary to interpret a large body of ongoing experiments on fuel production from water, and establish design rules to predict earth-abundant, non-toxic oxides with interfacial properties optimally suited to oxidize water.